Internal R&D Versus Externalization: Which Pharma Strategy Yields Greater Success?
Small Giants of Innovation
Over the last decade, small and mid-sized pharmaceutical organizations (SMIDs) have emerged as the primary drivers of innovation, responsible for the discovery of more than 70% of new molecular entities (NMEs) approved by the US Food and Drug Administration. Moreover, annual NME approvals have steadily increased since the R&D productivity crisis of the early 2000s, with 159 NMEs approved in the 2018-20 period compared to only 76 in 2009-11.
In this time frame, the smallest biopharma companies by market cap have truly earned the moniker ‘small giants of innovation.’
Molecules originating in this cohort have shown a CAGR of 8.4%, compared to 5% and 5.2% for large and mid-sized pharma. Fueled by high innovation in small company pipelines, overall pharma research productivity has risen significantly since the grim first decade of this century. Industry-wide, total R&D spend per FDA-approved NME decreased from $8.9Bn in 2009-11 to approximately $3.7Bn in 2018-20.
Renewed Emphasis on Retaining Value
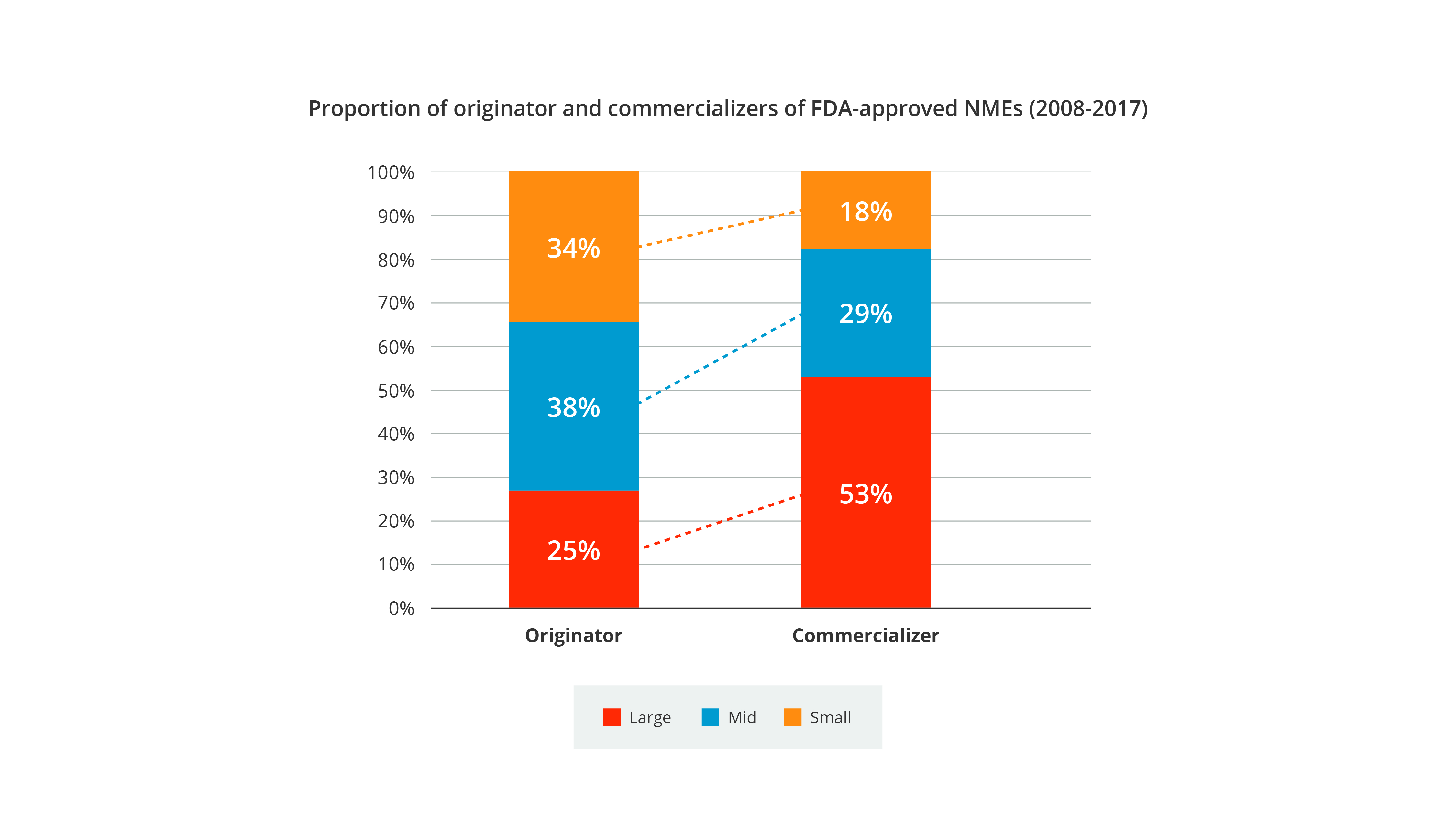
Some aspects of large-small pharma synergy have not changed. Large pharma companies have remained the engines of NME commercialization, with external R&D from SMIDs vitally augmenting large-cap pharma pipelines. Some 60% of NMEs originate in this manner.
However, in recent years, small and mid-sized companies have shown an increased appetite for in-house launches, building internal capabilities and outsourcing to enable commercialization while retaining higher shares of value. The trend for retaining value has intensified since 2018, marking a return to the levels of internalized versus externalized R&D seen in the early 2000s.
Internal R&D Advantage
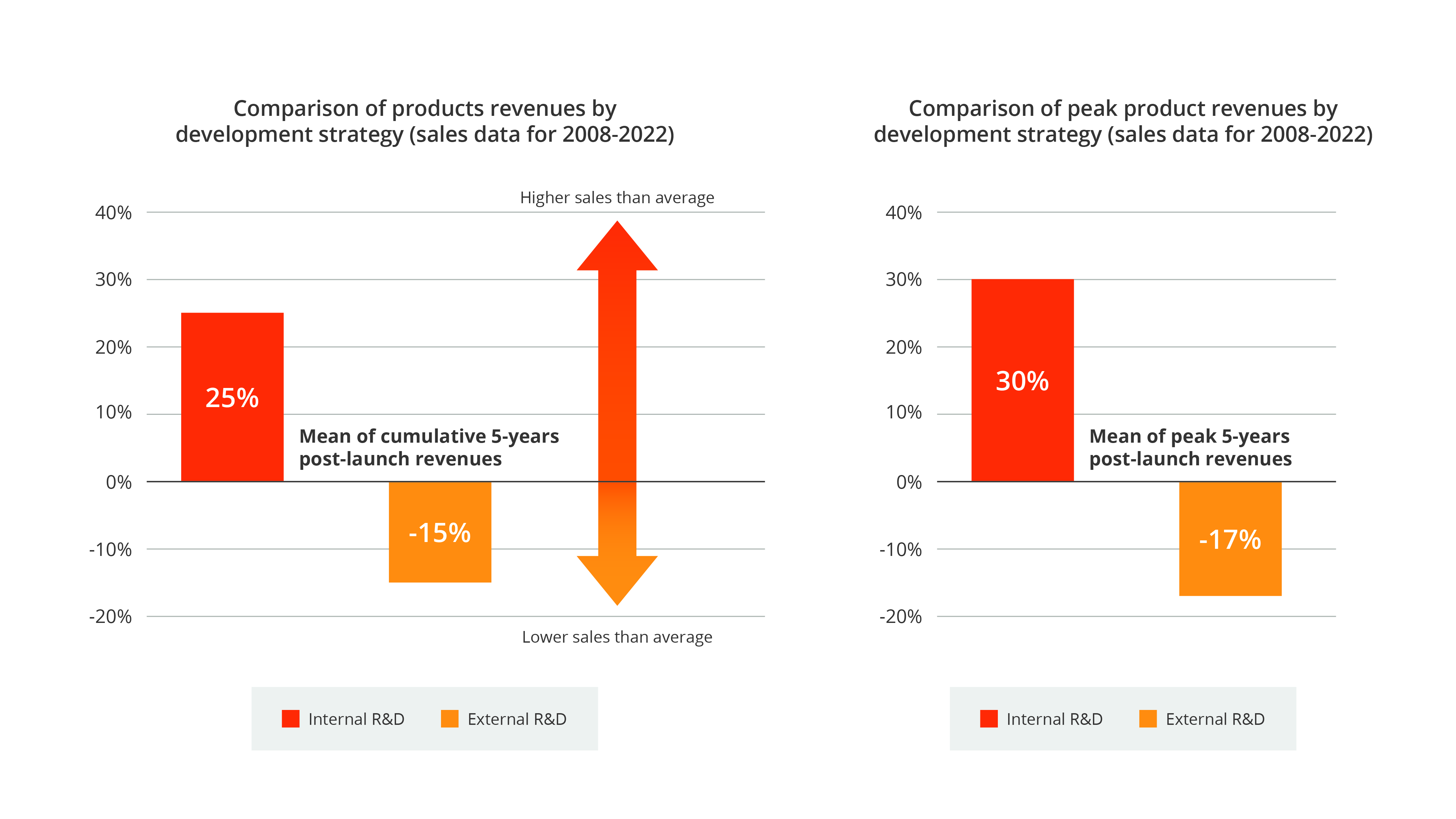
While large pharma’s reliance on external R&D from smaller biotechs is well recognized, industry experts have rarely compared the impact different innovation strategies (internal versus external) have on commercial success. Syneos Health® analysis of more than 15 years of NME innovation reveals that internally developed drugs are, on average, 40% more commercially successful than externally developed comparators.
As illustrated in the next two charts and additional data in the slideshow below, our thesis challenges the conventional wisdom that external R&D is a home run for pharma across the board. Our data underscores the commercial importance of maintaining deep in-house innovation from internal R&D while augmenting pipelines with external R&D. Internal small-company innovation and independent or assisted launches are increasingly leading to higher returns and creating opportunities to retain and maximize value.
Key takeaways from our analysis of 20 years of innovation:
- Small and mid-sized organisations have emerged as the innovation engine for pharmaceuticals, driving >70% of NME pipeline assets over the last decade
- While large pharma has remained the shepherd for taking NMEs to market, small and mid-sized companies have shown an increased appetite for in-house launches in the last 5 years
- External R&D has been responsible for >60% of NMEs, augmenting commercial pipelines and enabling large-cap pharma to bring NMEs to patients
- Internally developed NMEs have returned up to 40% more commercial value than externally developed NMEs on average in recent years
Syneos Health Consulting is committed to empowering clients daily by optimizing the value of their developed or acquired assets and facilitating both buy-side and sell-side M&A.
For more detail on the information presented above please see the detailed slides here and contact us for more information.
Contributors
Paul Holmes, Managing Director, Commercial Advisory Group, Syneos Health
Harry Bickerstaffe, Senior Consultant, Commercial Advisory Group, Syneos Health
Gursharan Batra, Associate Consultant, Commercial Advisory Group, Syneos Health
Larisa Djuric, Research Analyst, Info Access Data Intelligence, Syneos Health


